Mannitol: Unwrapping the Everyday Science Behind a Common Sugar Alcohol
Historical Development
Looking at the history of mannitol, it comes clear that this isn’t a newcomer to human applications. Folks discovered mannitol in plants centuries ago, isolating it from tree sap and seaweed. Its name traces back to manna, that sweet stuff described in old biblical texts. For a long time, scientists took advantage of its sweetening ability and natural presence in olives, figs, and mushrooms. Chemists later learned to separate it from other plant substances, like sorbitol, using extracted seaweed and then, more efficiently, by hydrogenating fructose. Mannitol's use shaped up quickly in the mid-20th century, responding to medicine's need for a sugar that doesn't spike blood sugar. European and American scientists published widely on its safety and the surprising range of things it could do, from sweetening foods for diabetics to drawing water out of swollen tissues in emergency rooms and clinics.
Product Overview
Mannitol stands as a sugar alcohol widely used both in food and pharmaceuticals. People recognize it in chewing gums labeled "sugar-free," and doctors reach for its IV drip when managing head trauma or high eye pressure. Unlike basic table sugar, mannitol slips under the radar of oral bacteria, so teeth don’t suffer as much. It runs through the kidneys without breaking down into glucose, which set it apart as a safer option for diabetics and folks struggling with metabolic disorders. As a powder, it looks like regular table sugar, white and crystalline, quick to dissolve and familiar in texture.
Physical & Chemical Properties
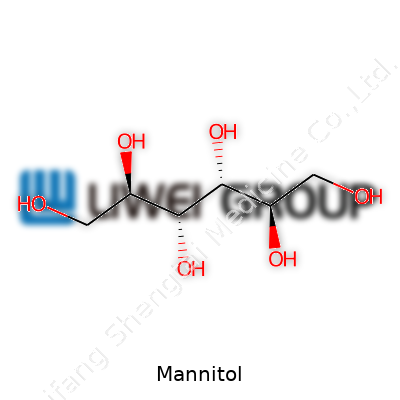
Mannitol falls into a growing family of polyols—sugar alcohols with a molecular formula of C6H14O6. If you placed it next to table sugar, most people couldn’t tell them apart by sight alone. Its melting point streaks past 165°C, and it resists absorbing moisture in the air, making it a stabilizer for medicines and candies alike. In water, mannitol tastes lightly sweet, nearly half as sweet as sucrose, but without the same glycemic punch. Chemically, each molecule has six carbon atoms and six hydroxyl groups, giving it solubility and stability under a range of conditions, including the high heat required for pharmaceutical processing.
Technical Specifications & Labeling
Looking over most commercial packages, the degree of purity makes a difference. Mannitol labeled as USP or PhEur grade refers to formulations meeting strict pharmaceutical quality standards, while food-grade versions might relax the rules a bit. Nutrition panels must show mannitol content, especially in regions where sugar alcohols trigger digestive discomfort at higher doses. Food safety laws in the United States and Europe cap the amount used in chewing gum and sugar-free snacks, reflecting past reports of gas and bloating at levels over 20 grams per day. It may show up under several approved names, but manufacturers must declare it for anyone tracking intake or allergies.
Preparation Method
Industrial teams make most mannitol by hydrogenating fructose, sourced from starch-heavy crops like corn or wheat. The process involves catalytic conversion, where producers pump hydrogen gas through liquid fructose with a nickel catalyst, transforming it through chemistry. The result is a mixture of sugar alcohols, and technicians separate out mannitol by applying differences in crystallization or by distillation. It’s a far cry from collecting sap or boiling seaweed resin—people everywhere rely on the efficiency of this modern method to meet the global demand, especially in the pharmaceutical industry.
Chemical Reactions & Modifications
Mannitol rarely stays static in laboratories. Organic chemists transform it into specialty surfactants by attaching different alcohol or acid groups. In some cases, they oxidize one of its hydroxyl groups to turn it into mannitic acid or create phosphate esters used in drug synthesis. Researchers also use mannitol as a starting point for designer molecules, tweaking its backbone to build biodegradable polymers or as carriers for drug delivery. These modifications don’t just stay in textbooks—they fuel ongoing research into new medicines, food science, and material chemistry.
Synonyms & Product Names
Supermarket buyers might miss mannitol in ingredient lists, because companies hide it behind its INS number 421, or call it manna sugar in the natural products aisle. In medical contexts, practitioners know it better as Osmitrol, especially when delivering it intravenously. In different regulatory zones, the name might switch, but the underlying molecule remains the simple, white powder that shows up in tablets, candies, and ice cream.
Safety & Operational Standards
Handling mannitol doesn’t usually spark safety worries, but good laboratory and manufacturing practices really matter. Regulatory bodies like the FDA and EFSA watch carefully, approving only specific concentrations for food and pharmaceutical use. Workers processing mannitol wear masks to avoid dust inhalation, especially given that fine powders irritate airways with chronic exposure. Storage in cool, dry places limits caking and contamination, so big operations lean on tight protocols. In hospitals, nurses and pharmacists stick to rigid procedures for preparing infusions, knowing that incorrect dosing could make fluids shift unpredictably in the body.
Application Area
Mannitol isn’t only a food additive or a sweetener for diet sodas. Doctors turn to it in emergencies, using it as an osmotic diuretic to reduce brain swelling after stroke or trauma, and to manage intraocular pressure in glaucoma attacks. It passes through the bloodstream without raising blood sugar, so diabetics find it in many processed foods labeled “low glycemic.” Looking past the pharmacy counter, it also pops up as a bulking agent in tablets, giving structure and volume without changing drug action. Candy makers use its cooling taste in mints and chewing gum, and bakers add it to keep washes shiny or maintain moisture. In biological research, mannitol preserves cells during freezing and protects sensitive proteins during industrial enzyme production.
Research & Development
Research teams keep pushing the limits of mannitol’s usefulness. Recent work explores how mannitol-based carriers could deliver cancer drugs right to tumors, releasing chemicals slowly and reducing side effects. Some biologists test it as a cryoprotectant, improving the survival of cells and tissues in organ transplants. Plant biologists have even tried feeding mannitol to crops under drought stress, hoping the sugar alcohol will help roots hold onto water better. These ideas spin out of ongoing studies in university and corporate labs, reminding us that even everyday chemicals like mannitol hide a universe of future innovations behind a plain appearance.
Toxicity Research
Plenty of studies focus on mannitol’s safety, and most people tolerate modest amounts without issue. Used as an IV drip, careful dosing remains essential, since too much can shift electrolytes and cause dehydration, especially in hospital patients with weak kidneys or unstable heart rhythms. In food, doses over 20 grams a day sometimes cause bloating or laxative effects—not surprising given its poor absorption from the gut. Regulatory panels have looked at birth defect rates, cancer studies, and long-term feeding trials, finding little evidence of harm at the amounts used in consumer products. For groups with special vulnerabilities, like children or people with rare metabolic disorders, doctors may suggest limits or alternatives.
Future Prospects
Looking forward, mannitol stands to be much more than a simple sugar substitute. Researchers tracking the global rise of chronic diseases see mannitol as a lifeline for diabetics or people managing metabolic syndromes. In the pharmaceutical world, scientists expect new drug-carrier systems and tissue-preservation methods to use mannitol in place of riskier or less stable chemicals. Food startups eye it as a way to lower calories without losing the familiar mouthfeel of conventional sugar, especially as regulatory pressure grows to reduce added sugar in processed foods. Environmental groups note the plant-based feedstock and relatively mild chemical processing, raising the possibility of a greener, more sustainable source for ever-growing demand. As science peels back the layers, mannitol’s story fits a pattern common in chemistry—taking something simple, often overlooked, and finding worlds of value hidden beneath each crystalline grain.
The Real Workhorse Behind the Scenes
Most folks hear “mannitol” and think of something out of a chemistry class. But those who have spent time with anyone with kidney trouble, brain swelling, or diabetes know it’s a workhorse in the hospital, not just a fancy label on a sugar-free snack. Mannitol is actually a sugar alcohol, found naturally in several plants, and yes, it gives that subtle sweetness to sugar-free gums and candies. But inside the hospital walls, it’s a different story. There, it quietly saves lives.
Getting Rid of Pressure Fast
Nothing rattles a room full of doctors quite like brain swelling. After a head injury, fluid can build up, and pressure starts to rise inside the skull. Mannitol isn’t the first thing most people reach for at home, but for those who go through a medical emergency, it can mean the difference between life and death. Given through an intravenous line, it draws water out of brain tissue, lowers pressure, and gives surgeons or medicines a fighting chance. If you’ve ever watched the worry drop from a neurologist’s face after a bad scan, you’d understand why mannitol holds a spot in the standard emergency kit.
Kicking Kidneys Into Gear
Problems with kidneys sneak up on people—one day you’re fine, the next your body fills with fluid, and nothing is leaving. Mannitol steps in there, too. In some hospital treatments, it acts as an osmotic diuretic. What that means for the patient is pretty simple: it pulls water into the kidneys so the body can flush it out. After surgery or an accident, doctors sometimes use mannitol to help wake up kidneys that have stalled, trying to nudge them back into action and keep toxins from piling up in the blood.
Beyond the Hospital Walls
Outside those high-drama uses, mannitol finds a home in the lives of people living with diabetes. Because it’s only partly absorbed by the gut, it sweetens food without causing the same spike in blood sugar as regular sugar. That’s a game-changer for anyone watching their glucose levels. I’ve seen folks with diabetes enjoy a piece of candy without a wave of guilt or worry, thanks to ingredients like mannitol. It’s not an everyday fix—eating too much brings stomachaches—but it’s still freedom in a small way for people who have to watch every bite.
The Double-Edged Sword
Problems pop up if people or doctors get too comfortable with mannitol. Too much can strain the heart or kidneys. It’s not for folks with severe kidney failure, since it lingers in the body. Children, older adults, and anyone with serious heart trouble need an expert hand guiding the dose. I’ve sat through shifts where a patient’s swelling dropped but their salt balance slid out of control, and the team scrambled to fix the new problem on top of the old one.
Room for Smarter Use
What keeps mannitol safe isn’t luck or clever chemistry—it’s the skill and judgment of the medical staff. Training and updated guidelines go a long way. More research into alternatives, better monitoring in real time, and patient education could cut down on risky side effects. If there’s one truth in medicine, it’s that no quick fix comes free. Mannitol proves its worth every day, but it keeps everyone on their toes.
Understanding Why Mannitol Packs a Punch
Mannitol sits on the list of medicines I once took for granted—until a close friend wound up using it in the hospital. Anyone who's seen it given for brain swelling or as a solution for kidney problems probably won't forget the swift reactions and the need for careful monitoring. Mannitol tends to work fast and does what doctors ask, but it can cause a laundry list of side effects if not used with caution. Anyone thinking this is just sugar water will be in for a surprise.
Kidneys, Fluids, and the Balancing Act
One thing stands out about mannitol: it pulls water out of tissues and into the bloodstream. This is a lifesaver for people with certain brain injuries or those who need their kidneys pushed into action. Yet as this water makes its way out, patients can lose not just fluid but also important body salts, like sodium and potassium, in the urine. I once watched a patient’s potassium crash within hours. Low potassium means weak muscles, irregular heartbeats, and a hospital team on edge. Hyponatremia, or low sodium, can bring confusion and even seizures. That’s real danger, not just numbers on a chart.
Dehydration: More Than “Just Thirst”
Some people assume dehydration just makes you thirsty. With mannitol, fluid leaves the body fast, sometimes faster than a person can drink water or the medical team can replace it. The skin gets dry, blood pressure drops, and the kidneys may suffer. It’s a cruel irony — a drug meant to help the kidneys can sometimes push them into failure, especially for patients already running on empty or those with heart issues. Hospitals should pay attention here, with regular lab tests and sharp clinical eyes. Nobody wants kidney shutdown because a line was running too quickly.
Allergic Reactions and Breathing Trouble
Though less common, allergic reactions aren’t impossible. Anyone with a history of allergies or asthma has to take extra care. Breathing trouble, rash, or even anaphylaxis can come on without warning. This isn't the place for hesitation—if someone starts wheezing or swelling up, pulling the plug and calling for help beats sticking to a schedule.
Brain Swelling and Rebound Risks
Doctors love mannitol for lowering brain pressure, but too much or too fast can lead to rebound swelling. The very problem being treated might come back, worse than before, once mannitol is stopped or wears off. Families put a lot of trust in medical choices at times like these, so the team’s experience and close follow-up matter more than ever.
Lessons from the Hospital Floor
Mannitol deserves respect. Teams on the ground know that regular lab checks, tracking urine, and listening to the patient’s complaints aren’t extra — they’re essential. Minor headaches and nausea may be the first clues that something’s wrong. Nonstop monitoring may sound like overkill, but it has saved lives.
Fixing Serious Problems Calls for More Than Protocols
Hospitals do best when they build teams that work together: the nurses who catch changes, the pharmacists who review doses, the doctors who steer the ship. Everywhere I’ve worked, open communication has made the difference. Health systems should push for this kind of teamwork and never assume mannitol is just another IV drip. Better training, quicker response to warning signs, and careful choices about which patients get it make all the difference.
Mannitol is more than a name on an IV bag in the hospital; for people dealing with brain swelling, glaucoma, or certain kidney conditions, mannitol is a literal lifesaver. The way it’s given can decide whether it helps or brings on new problems, which is something folks outside the medical field don't always realize. Hospitals keep this sugar alcohol on hand for good reason, but just having it within arm’s reach doesn't mean the job’s done.
Giving Mannitol Isn’t Like Swallowing a Pill
Doctors rarely reach for pills when mannitol is called for. Instead, this solution flows straight into the vein, usually through a slow, well-controlled intravenous drip. This matters because tossing too much into a person’s system too quickly can overwhelm the kidneys, mess with blood pressure, and set off a cascade of new complications. Patients in the emergency room trust us to use precision, so nurses monitor every drop and keep a close eye on vital signs while the infusion does its work.
From personal time spent on hospital wards, I’ve watched critical care teams gather for a quick huddle before starting mannitol. The reason? Timing, speed, and even the size of the IV needle can change the outcome for a patient. If a child or senior needs mannitol, the calculations shift, showing that real-life medicine always beats textbook theory.
Why Monitoring Matters With Mannitol
Every medical pro I know shares stories about watching someone’s brain swelling shrink after getting the dose just right; those moments stick with you. But the flip side—too much, too fast—can tip a vulnerable patient into kidney failure or dangerous electrolyte shifts. On many shifts, the pharmacy plays its own crucial part, mixing up mannitol solutions that stay sterile and stable. Nurses draw up clear solutions and look for any crystals in the bag, since mannitol turns cloudy in cold conditions, which signals risk if injected as is. Everyone from the pharmacist to the bedside nurse checks twice before a single drop goes in.
Challenges Clinicians See Every Day
Setting up an infusion sounds simple on paper, but it gets tricky in the heat of an emergency. Sometimes IV lines get blocked by mannitol crystals, setting off alarms in the ICU. Teams often use in-line filters to catch any particles, a step that protects patients and saves time in the long run. Distractions lead to mistakes, so protocols push for careful double-checking and patient-specific dosing. In real-world practice, teams face questions like: How dehydrated is this patient? How are their kidneys doing? These questions aren’t academic; lives swing on tiny details.
Better Systems Mean Safer Outcomes
I’ve seen hospitals invest in precise infusion pumps, staff training, and standardized guidelines to reduce mistakes. These systems can cut down on dosing errors and allow staff to respond fast to changing blood test results. Some smart hospitals have pharmacists join clinical rounds to spot problems before they start. That cross-checking approach stands between a risky guess and a life-saving decision.
Lessons For Patients and ProvidersA quick internet search won’t replace hard-won knowledge about giving mannitol safely. For patients, clear questions about the “how and why” of an IV treatment matter. For healthcare teams, it’s about respecting the details every time, knowing that a seemingly simple bag of fluid can change everything—for better or worse.
Looking at Mannitol in the Hospital Hallways
Mannitol shows up in hospitals everywhere, usually in IV bags marked for folks with brain swelling or raised pressure inside the eyes. It's a sugar alcohol that doctors trust to pull extra water out of body tissues and send it off for the kidneys to excrete. Some people call mannitol a “renal workhorse,” but in real-world practice, that job description only fits healthy kidneys. I’ve stood at the bedside, watching doctors decide if mannitol is safe to use, especially for folks whose kidneys are not doing their best work.
Why Folks With Kidney Issues Need a Second Look
Kidneys filter blood and help get rid of waste. If they’re a bit sluggish or flat-out failing, giving a medicine like mannitol can change the game. It draws water out of swollen tissues, but if those kidneys can’t dump the extra fluid fast enough, trouble starts. That excess liquid stays in the blood, swelling builds up, lungs get wet, and suddenly a patient developed breathing problems. I’ve seen mannitol turn from a helping hand into a real threat in patients sitting on the edge of kidney failure.
Looking at the Risks Up Close
Kidney specialists, both in the ICU and on the hospital floor, watch for mannitol’s rarely discussed side effect—osmotic nephrosis. Too much mannitol loads the kidneys with sugar-alcohol molecules they can’t process, leading to swelling inside the filtration system. It makes a bad problem worse: instead of helping, kidney function tanks even further. The Food and Drug Administration’s labeling warns doctors—it’s not for use in folks with established severe kidney damage or issues with anuria, which means absolutely no urine is coming out.
More than that, studies in critical care show outcomes dip when mannitol is used in folks with already-compromised kidneys. Some large trials in people with brain injuries didn’t show better outcomes than other medicines, such as hypertonic saline, for reducing brain swelling. Hospitals now try to check kidney function carefully before going to mannitol. Blood tests—like creatinine and estimated glomerular filtration rate—step in as guides. Doctors usually want numbers to look safe and stable before considering a single dose.
Are There Safer Alternatives?
Modern medicine doesn’t lean on mannitol as much as it used to. Newer strategies focus on using hypertonic saline, which seems less likely to cause problems for patients with kidney struggles. Hypertonic saline pulls water out of swollen tissues, just like mannitol, without putting as much strain on kidneys. In my own practice, teams sometimes skip mannitol altogether if bloodwork suggests even mild kidney injury.
What Should Be Done?
For patients with kidney issues, teams need to sit down and look at the big picture. Is the brain swelling bad enough to risk the kidneys? Will mannitol offer enough benefit to outweigh the dangers? Every dose brings that question to the front of the room. If a patient already has kidney disease or is teetering on the edge of failure, medical teams often look for another option. That approach doesn’t just save kidneys—it can keep folks out of the ICU and even save their lives.
Facts and experience both point in the same direction. Mannitol can be helpful, but for those who walk into the hospital with weak kidneys, the risks usually run too high. It’s not just about numbers on a lab sheet; it’s about doing right by patients by picking the medicine least likely to cause harm.
Mannitol’s Place in Medicine
In hospitals, Mannitol often steps in to reduce swelling in the brain or to kickstart kidney function in patients facing acute kidney trouble. On paper, it looks like a miracle. But anyone who's seen Mannitol at work knows it walks a thin line between life-saving and risky. Respect for this drug comes naturally if you’ve ever watched a patient’s labs veer dangerously out of range after a dose.
Why Mannitol Calls for Close Attention
What makes Mannitol tricky is its ability to tug water from the body’s tissues and send it into the bloodstream. This sudden shift lowers pressure in the brain or eye, but folks with heart or kidney problems can end up with more fluid in their blood than their bodies can handle. I once watched a man’s lungs fill with fluid because his heart couldn’t keep up — a tough reminder that medicine’s power always comes with a catch.
Where Mannitol Can Go Wrong
Some people look fine after a single IV, but the drug’s habit of pulling electrolytes along with water means sodium and potassium can plummet or spike. Those swings can set the stage for confusion, muscle weakness, or heart rhythm problems. People with kidney or heart conditions start with less wiggle room, so doctors often double-check blood tests before giving a dose.
The body’s reaction doesn’t always follow the rule book. After years on the medical floor, I’ve learned never to trust routine, especially with medications like Mannitol. Even if someone took it last year with no issue, a new infection, a fresh heart problem, or bad dehydration can tip the balance.
What Caution Looks Like in Real Life
A person getting Mannitol usually ends up with an extra monitor or two: checking blood pressure, watching for crackles in the lungs, and drawing blood to catch electrolyte swings early. It’s not wasted effort. One missed sign can turn a cautious treatment into a dangerous setback. I’ve seen experienced nurses walk through the room, eyes trained for swelling, confusion, cough, or too much thirst. These simple checks catch trouble earlier than any computer alert.
Labs tell a story, but so does common sense. Mannitol tends to move fast, and so should the team around it. Good practice involves having repeat blood tests queued up, extra IV fluids only if salt levels drop, and holding back on the next dose if there’s any sign kidneys aren't flushing out the drug as expected.
Smarter Prescribing and Patient Awareness
Medical teams keep Mannitol as an option, not a go-to. Only careful conversation between doctor, nurse, and patient decides if the benefits really outweigh the risks, especially with people already in the danger zone for heart or kidney problems. For patients, asking questions about hydration, side effects, and what to expect helps them stay part of the process.
Pharmacy checks make a big difference. Mixing up the dose, or pushing it too fast, lands patients in trouble. Having pharmacy and bedside teams both check orders cuts those mistakes way down. Simple steps like clear labeling and time-outs before administration can mean the difference between smooth care and an avoidable crisis.
Learning from Real-World Experience
Looking back, I’ve never met a medicine without risk. Mannitol’s power isn’t the problem; forgetting its risks is. What keeps patients safe is careful teamwork, slow observation, respect for changing lab results, and listening to what patients say about how they feel. It’s not high-tech — it’s about experience, clear eyes, and never letting routine make us careless.